Key Features
EC™ Portfolio
Hemodynamic monitoring is of great importance in all hospital departments, and almost on all patients. In order to cover this wide range of applications and to facilitate the interpretation of a hemodynamic condition, the Electrical Cardiometry™ (EC™) portfolio offers various special features.
Electrical Cardiometry™ Portfolio includes the ICON® and ICONCore®. Each device has its own features by sharing the same technology.
ICON® is a small hand-held and thus portable device especially suitable for mobile use.
ICONCore® together with iControl™ offers a comprehensive user interface and extended functions.
| ICON® | ICONCore® + iControl™ Version 4.x |
|
| Parameter Spectrum | SVR after specification of blood pressure and DO2 after input of SpO2 and Hb (manual entry) | SVR after specification of blood pressure and DO2 after input of SpO2 and Hb (manual entry or LAN (WiFi)) |
| Data Acquisition | Beat-to-beat or average |
Beat-to-beat or average |
| Hemoview™ Diagnostic Bar Screen | ||
| Trend Screen | ||
| Hemodynamic Status Reports | Single or comparative | |
| PLR Test Guidance | ||
| Therapeutic Management Screen | ||
| Input of Blood Pressure | Manual entry | Manual Entry & LAN (WiFi) |
| Export Input of SpO2 & Hb | Manual entry | Manual Entry & LAN (WiFi) |
| Export Measurement Records (numerical values) |
Yes, depending on recording period (default: 1 min) and averaging mode (default: 60 cardiac cycles); ASCII file format | Yes, beat-to-beat to a selectable recording period; selectable averaging; CSV file format |
| Battery Operation (full charge) | ≥ 2 h | ≥ 4 h (PC not considered) |
| External Communication | HL7, Philips Vuelink / Intellibridge, Data Logging | HL7 |
Key Features explained
Parameter Spectrum
Electrical Cardiometry™ devices provide a wide range of hemodynamic parameters:

Blood Flow:
- Stroke volume (SV) and stroke index * (SI)
- Cardiac output (CO) and cardiac index * (CI)
- Heart rate (HR)
Fluid Status:
- Stroke volume variation (SVV),
- Corrected flow time (FTc),
- Thoracic Fluid Content (TFC)

Contractility & Cardiac Function:
- Contractility Index (ICON),
- Systolic Time Ratio (STR),
- Pre-ejection period (PEP),
- Left Ventricular Ejection Time (LVET)
- Cardiac Performance Index (CPI)
- Variation in contractility (VIC)
Vascular Resistance:
- Systemic vascular resistance (SVR) and its index * (SVRI)
(ICON® & ICONCore®: after entry of blood pressure value)

Blood Pressure:
- Mean arterial pressure (MAP),
- Systolic arterial pressure (SBP),
- Diastolic arterial pressure (DBP)
(Manual Entry & LAN (WiFi))
Oxygenation:
- Supplied oxygen or its index * (DO2 / DO2I),
- Blood oxygen saturation (SpO2),
- Hemoglobin content (Hb)
(ICON® and ICONCore® after specification of SpO2 and Hb)
(* Indices can be related to body surface area or weight)
Data Acquisition
Recording settings can be easily adjusted for all EC™ devices. The averaging depth is selectable in specific steps, starting from five seconds up to three minutes or from five cycles up to 180 cycles. Even a precise beat to beat recording is possible.
The setting of variation parameter range can take place in easy steps between 10 cycles and up to 60 cycles. Parameters can be indexed to body surface area or to weight.
Electrical Cardiometry™ can therefore be used in everyday clinical practice on all patient groups and is also suitable for precise analysis and research purposes.
Hemoview™ Diagnostic Bar Screen
![]() Hemoview™ is a unique diagnostic display available on all EC™ devices. It enables quick interpretation of a patient’s condition.
Hemoview™ is a unique diagnostic display available on all EC™ devices. It enables quick interpretation of a patient’s condition.
Parameter values are graphically represented by a colored bar that relates to the normal range of each individual patient (based on the demographic data entered at the beginning of a measurement). In addition, numerical values are specified.
Trend Screen
The value of the parameter over time is visualized in the trend screen. Changes in hemodynamics can be assessed over time and therapies carried out and tracked accordingly. A marker function simplifies detecting response for medical interventions.
The trend display and marker function are available on all EC™ devices.
Hemodynamic Status Report
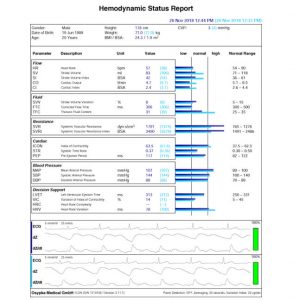 A status report represents the hemodynamic status at a specific point of time. Status reports can be recorded at any time and saved in PDF format.
A status report represents the hemodynamic status at a specific point of time. Status reports can be recorded at any time and saved in PDF format.
Parameters are sorted according to their hemodynamic affiliation and an overall interpretation is simplified using the Hemoview™ (bar) chart.
Changes in the hemodynamics can be compared using several status reports or directly compared using a comparison report. Hemodynamic changes, such as therapy effects, can be seen immediately. The signal quality is always indicated on the basis of raw data and a percentage display. Parameter normal ranges, depending on the given patient data, are listed.
Hemodynamic status reports can be created for all EC™ devices. Comparison reports can be generated with the iControl™ software, available at ICONCore®.
PLR-Test Guidance
The Passive Leg Raise Test (PLR) is a bedside determination of volume responsiveness according to Frank Starling law. It requires continuous CO measurement.
Starting from the semi-recumbent position (upper body 45° upright), the patient is passively brought into a lying position and the legs raised by 45°. Driven by gravity, venous blood moves from the legs towards the heart. This increases the preload (blood volume returning to the heart) for a short time.
If a patient reacts positively, the cardiac output increases by at least 15%, signifying the patient is fluid responsive.
A protocol implemented in iControl™ allows a guided execution of a PLR test with subsequent specification of the patient’s volume responsiveness. The software iControl™ is used with ICONCore®.
Therapeutic Management Screen
The pure consideration of the stroke volume and the mean arterial blood pressure, including their normal ranges, provides initial information for therapeutic approaches with regard to the cardiovascular system. The current status is continuously visualized in the therapeutic management screen.
Noninvasive Blood Pressure
ICONCore® offers the possibility of integrating blood pressure via LAN (WiFi).
Manual entry of the NIBP is possible with all EC™ devices. The blood pressure is used to determine the systemic vascular resistance.
Oxygenation
To determine DO2, SpO2 and Hb can be entered manually.
Data Export
Parameter data is provided numerically. The ICON® saves parameter values depending on the defined recording interval (default is 1min) and the averaging depth (default is 60 cardiac cycles).
iControl™ allows numerical parameter data to be configured before an export. A specific time window can be set, the recording interval and the averaging depth can be redefined and an exclusion limit for the signal quality index (SQI) can be set. If necessary, it is also possible to select only data points with marker entries.
Data from PLR tests are provided separately. iControl™ is used with ICONCore®.
Battery Operation
In battery mode, the ICON® can be used for at least two hours, the ICONCore® for at least four hours.
Therefore, it is possible to operate all EC™ devices on the move.
External Communication
ICON® and ICONCore® are equipped with an HL7 interface, which allows parameter data to be transferred to a patient data management system (PDMS).
ICONCore® also enable a PDMS query for the continuous integration of external parameters.
In addition, the ICON® offers data logging as well as a unidirectional interface to Vuelink™ / Intellibridge™ technology.
